6.3 The pH Scale
Learning Objectives
- Define pH.
- Determine the pH of acidic and basic solutions.
As we have seen, [latex]\ce{[H^{+}]}[/latex] and [latex]\ce{[OH^{-}]}[/latex] values can be markedly different from one aqueous solution to another. So, chemists defined a new scale that succinctly indicates the concentrations of either of these two ions.
pH is a logarithmic function of [latex]\ce{[H^{+}]}[/latex]:
[latex]\text{pH}=-\log[\ce{H+}][/latex]
pH is usually (but not always) between 0 and 14. Knowing the dependence of pH on [latex]\ce{[H^{+}]}[/latex], we can summarise as follows:
- If pH < 7, then the solution is acidic.
- If pH = 7, then the solution is neutral.
- If pH > 7, then the solution is basic.
This is known as the pH scale. You can use pH to make a quick determination of whether a given aqueous solution is acidic, basic, or neutral.
Example 6.3.1
Problem
Label each solution as acidic, basic, or neutral based only on the stated pH.
- milk of magnesia, [latex]{pH = 10.5}[/latex]
- pure water, [latex]{pH = 7}[/latex]
- wine, [latex]{pH = 3.0}[/latex]
Solution
- With a pH greater than 7, milk of magnesia is basic. (Milk of magnesia is largely [latex]\ce{Mg(OH)_{2}}[/latex].)
- Pure water, with a pH of 7, is neutral.
- With a pH of less than 7, wine is acidic.
Test Yourself
Identify each substance as acidic, basic, or neutral based only on the stated pH.
- human blood, [latex]{pH = 7.4}[/latex]
- household ammonia, [latex]{pH = 11.0}[/latex]
- cherries, [latex]{pH = 3.6}[/latex]
Answers
- basic
- basic
- acidic
Table 6.3.1 “Typical pH Values of Various Substances” gives the typical pH values of some common substances. Note that several food items are on the list, and most of them are acidic.
| Substance | pH |
|---|---|
| stomach acid | 1.7 |
| lemon juice | 2.2 |
| vinegar | 2.9 |
| soda | 3.0 |
| wine | 3.5 |
| coffee, black | 5.0 |
| milk | 6.9 |
| pure water | 7.0 |
| blood | 7.4 |
| seawater | 8.5 |
| milk of magnesia | 10.5 |
| ammonia solution | 12.5 |
| 1.0 M NaOH | 14.0 |
pH is a logarithmic scale. A solution that has a pH of 1.0 has 10 times the [latex]\ce{[H^{+}]}[/latex] as a solution with a pH of 2.0, which in turn has 10 times the [latex]\ce{[H^{+}]}[/latex] as a solution with a pH of 3.0 and so forth.
Using the definition of pH, it is also possible to calculate [latex]\ce{[H^{+}]}[/latex] (and [latex]\ce{[OH^{-}]}[/latex]) from pH and vice versa. The general formula for determining [latex]\ce{[H^{+}]}[/latex] from pH is as follows:
[latex][\ce{H+}]=10^{-\text{pH}}[/latex]
During your studies, it will be necessary to evaluate the above expression on your calculator. This will not be covered in this book; watch this video to learn how to perform pH calculations using a calculator.
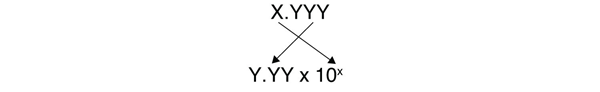
The other issue that concerns us here is significant figures. Because the number(s) before the decimal point in a logarithm relate to the power of 10, the number of digits after the decimal point is what determines the number of significant figures in the final answer, as you can see below:
Example 6.3.2
Problem
What are [latex]\ce{[H^{+}]}[/latex] and [latex]\ce{[OH^{-}]}[/latex] for an aqueous solution whose pH is 4.88?
Solution
We need to evaluate the following expression:
[latex][\ce{H+}]=10^{-4.88}[/latex]
Depending on the calculator you use, the method for solving this problem will vary. In some cases, the “−4.88” is entered and a “10x” key is pressed; for other calculators, the sequence of keystrokes is reversed. In any case, the correct numerical answer is as follows:
[latex][\ce{H+}]=1.3\times 10^{-5}\text{ M}[/latex]
Because 4.88 has two digits after the decimal point, [latex]\ce{[H^{+}]}[/latex] is limited to two significant figures. From this, [latex]\ce{[OH^{-}]}[/latex] can be determined:
[latex][\ce{OH-}]=\dfrac{1\times 10^{-14}}{1.3\times 10^{-5}}=7.7\times 10^{-10}\text{ M}[/latex]
Test Yourself
What are [latex]\ce{[H^{+}]}[/latex] and [latex]\ce{[OH^{-}]}[/latex] for an aqueous solution whose pH is 10.36?
Answer
[latex]\ce{[H^{+}]}[/latex] = [latex]{4.4 × 10^{-11} M}[/latex]; [latex]\ce{[OH^{-}]}[/latex] = [latex]{2.3 × 10^{-4} M}[/latex]
There is an easier way to relate [latex]\ce{[H^{+}]}[/latex] and [latex]\ce{[OH^{-}]}[/latex]. We can also define pOH in a similar way to pH:
[latex]\text{pOH}=-\log[\ce{OH-}][/latex]
In fact, p“anything” is defined as the negative logarithm of that anything. This also implies that:
[latex][\ce{OH-}]=10^{-\text{pOH}}[/latex]
A simple and useful relationship is that for any aqueous solution:
[latex]\text{pH}+\text{pOH}=14[/latex]
This relationship makes it straightforward to determine pH from pOH or pOH from pH and then calculate the resulting ion concentration.
Example 6.3.3
Problem
The pH of a solution is 8.22. What are pOH, [latex]\ce{[H^{+}]}[/latex], and [latex]\ce{[OH^{-}]}[/latex]?
Solution
Because the sum of pH and pOH equals 14, we have:
[latex]8.22+\text{pOH}=14[/latex]
Subtracting 8.22 from 14, we get:
[latex]\text{pOH}=5.78[/latex]
Now, we evaluate the following two expressions:
[latex]\begin{array}{rcl} \left[\ce{H+}\right]&=&10^{-8.22} \\ \\ \left[\ce{OH-}\right]&=&10^{-5.78} \end{array}[/latex]
So:
[latex]\begin{array}{rcl} \left[\ce{H+}\right]&=&6.0\times 10^{-9}\text{ M} \\ \\ \left[\ce{OH-}\right]&=&1.7\times 10^{-6}\text{ M} \end{array}[/latex]
Test Yourself
The pOH of a solution is 12.04. What are pH, [latex]\ce{[H^{+}]}[/latex], and [latex]\ce{[OH^{-}]}[/latex]?
Answer
pH = 1.96; [latex]\ce{[H^{+}]}[/latex] = 1.1 × 10−2 M; [latex]\ce{[OH^{-}]}[/latex] = 9.1 × 10−13 M
Watch the following simulation about the pH scale.
Key Takeaways
- pH is a logarithmic function of [latex]\ce{[H^{+}]}[/latex].
- [latex]\ce{[H^{+}]}[/latex] can be calculated directly from pH.
- pOH is related to pH and can be easily calculated from pH.
Exercises
- Actual values may vary depending on conditions. ↵
A logarithmic scale shows the base value of 10 raised to the power of a value. For example, 10 has a logarithm of 1 because 10 raised to the power of 1 is 10. 100 has a logarithm of 2 because 10 raised to the power of 2 is 100.

